-> My Collection; No source to show as I've not shared the Mining Journal as an album.
But, here is a link to first page on Hathi Trust Digital Library website.
August, 1899 (page 5->7)
I've added the colored image as that was not part of the article source.
There are some mills and works that offer to the visitor points of both scientific and spectacular interest such, for example, as a Bessemer steel works and big rolling mill, and such also are some of our large typical cyanide mills at Cripple Creek.
We propose to ask such of our readers as may not be familiar with mills of this class to accompany us, following, step by step, the various processes through which the ore passes from its arrival at the mill till it leaves the zinc shavings of the precipitating boxes, and is handed over to the smelting furnace to be turned into a gold brick ready for the mint.
It was early found in the case of Cripple Creek, that some process on the ground was necessary to treat the vast amount of low grade ores that would not bear the expense of distant transportation and smelting. Ordinary stamp mills failed to meet the exigency; recourse was therefore had to the cyanide process.
The ores to be treated were those impregnating the country rock of altered andesite, phonolite, and granite, viz., iron oxides, carrying free gold, unoxidized iron pyrites low grade in gold, and tellurides rich in gold. Associated with these were quartz and fluorite.
The brown oxidized ores were found directly amenable to cyanide, but the bluish-gray, unoxidized ores required roasting before yielding a satisfactory extraction, the tellurides being all but insoluble in 0.5 per cent. cyanide. By roasting, the tellurium is oxidized and the gold set free in the metallic state, easily soluble in the ordinary cyanide solutions.
Three cyanide mills were accordingly erected, at different times, in the vicinity. The Brodie mill near Mound City, the Colorado Ore Reduction Company's works, or Arequa mill, in Arequa gulch, and the Metallic Extraction Company's mill at Cyanide, near Florence, the latter being the largest and most elaborate. For the purpose of this article, viz., that of giving an idea of an average cyanide mill, we will select that of the Colorado Ore Reduction Company's works.
This mill is situated on the steep slope of Arequa gulch, the individual buildings, representing often different stages in the process, are all united and descend in terraces to the foot of the hill and to the bottom of the gulch. The topmost building is a shed, open at both ends, into which the big ore wagons from the mines, drawn by four or six horses, enter, discharge their ore into bins allotted them, and pass out in succession.
We will now follow the ore in its progress down through the mill, from floor to floor, and process to process.
As the ore on arriving at the mill is of all shapes and sizes, after passing through the bins, the first process is to crush it preparatory to sampling it.
Below the bins is a tramway, on which a small car receives the crude ore as it issues from the chutes in the bottom of the bins, and wheels it to a grizzly, with bars, three-fourths of an inch apart and forty-eight inches long. What passes between these bars goes to the Blake crusher, while the larger pieces are broken by a sledge hammer, and then sent through to the crusher with the rest, and into the same bin, in the upper portion of the building.
The floor of the hand-sampling room is covered with broad steel plates, swept bright and clean. Small bins occupy one side of the room, containing ore of different shades of color and quality; a few long-bladed shovels and a hand sieve complete the equipment.
The ore from the crusher and the rolls on the bin floor below, after being elevated, is run through a Vezin automatic sampler, which takes a sample of the ore, and this sample is then taken to the hand-sampling room where it is still further reduced in bulk by quartering.
A wheelbarrow load of ground-up ore, which has been taken by the Vezin sampler, is dumped in the middle of the floor and two men with the sampling shovels take shovelfuls from every part of this heap and form a second smaller cone. This process is called "coning." The second process is "spreading" this cone by a peculiar and rapid circular motion of the shovels until the heap is resolved into a flat spiral mass; this is quartered, two quarters being thrown to the bins, and the two opposite quarters again coned and requartered, till the sample is small enough to be taken to the sampling office adjoining. Here the ore is dried in a small compartment lined with hot steam pipes, and the percentage of moisture determined. Thence it passes through a small Davis roll, and from that to a machine, appropriately termed the "coffee mill," where it is still further reduced. Thence it passes to the buckboard and maul. Samples are taken by the usual split-shovel sampler. On a white rubber sheet the sample is thrown and thoroughly mixed. From this the control assay samples are selected, and sealed up in small paper sacks made expressly for this purpose.
On the same landing is a long room, 100 feet by 30 feet, with piles of different colored ore on one side, and hoppers full of the same on the opposite. The brown ores are the oxidized ones, which, after crushing, go direct to the cyanide vats, without preliminary drying or roasting. The other ore, of a blue-grade tint, and full of unoxidized pyrite and some tellurides , has to pass through all the preliminary processes of drying and roasting before it can be treated either by the cyanide or chlorination process. The ore on this floor has been crushed to about the size of very coarse gravel. We open a door on the side of the room and look down into a noisy and dusty compartment where the screens are at work and elevators are carrying the material to the crushing rolls.
Ascending a ladder from this floor we find ourselves in a small upper room, in which the Vezin automatic sampler is revolving rapidly, with its four hollow arms, or "flights," as they are called, full of ore caught from a shower descending from above. What is caught in these flights is the "sample" and this goes down through a 6-inch pipe to the sampling floor below; what misses or passes between the flights goes down into a hopper and chute to a bin directly beneath, whence it is wheeled to one of the general storage bins. From these bins the ore is drawn into cars and taken to the dryer. Descending to the floor again, we notice a pile of ordinary burned lime; this is crushed and mixed in the cyanide tanks with the ore, in order to neutralize its acid character.
Proceeding to the next floor below, a tramway leads through a narrow alley lined on both sides with rows of chutes discharging comminuted ore received from the rolls above into a car on the track, which takes it and dumps it into the dryer.
There are three sets of rolls of solid iron, jacketed with steel; two are for the cyanide, and one for the chlorination, process. Alongside of the crushing department is a room quite empty, except for a large pile of dust lying in the center, which is being fed from two spouts, descending from the screen room above. This refuse dust will later be cyanided, and contains values. Of the screens, there are six used for cyanide, and two for chlorination material. The cyanide screens are 20-mesh; those for chlorination are 12-mesh.
From this floor we descend to that on which the revolving dryers are located. These consist of two large, hollow steel cylinders, 20 feet long by 4 feet in diameter, revolving slowly, side by side, with their burden of drying ore. One dryer is for chlorination, the other for cyanide ore. These cylinders taper slightly towards the end farthest from the furnace. They are divided lengthwise into four partitions, like a child's kaleidoscope, and this partition structure assists in exposing the ore to a drying heat. The dryers are fed with ore by an automatic feeder. The furnace that supplies the heat is at one end of the cylinder, and is fed by burning oil, sprayed by a steam jet. The dried matter from the cylinder is carried by an elevator, close by, up to rolls, and again elevated to screens. The screened ore is carried, by a screw conveyor, to the pulp bins, located just above the roasting furnaces, to be trammed either to the roasting furnace, or, in case of the oxidized ore, direct to the cyanide vats.
Outside this building is the dust chamber connected with the furnace inside by a big metal pipe, four feet in diameter. This dust is cleaned out at intervals and cyanided, as it carries a percentage of value.
On the same floor as the dryers are three boilers and a 120-horsepower engine adjoining. Following the track of a tramway it leads us into the long room occupied by the Ropp roasting furnace, conveyors, and cyanide tanks.
This is a weird-looking region, especially at night, of fire and heat and uncanny machines called "rabbles," crawling slowly along, one after another, about fifty feet apart, in a slow, ceaseless procession. The rabble is a long, thick steel bar, to the lowest surface of which are attached plates or blades. This bar with teeth is attached to a triangle of iron, bound together by long, flat iron bars, which are curved up at the ends. The machine travels on a small truck fitting on to the rails down the long tramway, around the side and ends of the furnace, drawn by an endless revolving wire rope, connecting the rabbles with one another. So the procession crawls along till a turntable is reached, opposite the far end of the furnace.
Let us follow the rabble in its passage through the furnace. On passing through the first set of flap doors, it presently encounters a similar set which again close after it, so as to exclude any draft let in by opening the first set. Inside the furnace the rabble finds the ore lying in a heap fed by an automatic and regulated hopper from above. This accumulated ore the rabble catches up, and with its harrow-like teeth distributes along its path on the floor of the furnace. The succeeding rabble, with teeth set at a different angle, turns over the work of its predecessor in an opposite direction, so that eventually every portion of the material is well worked over, and receives its full share of heat.
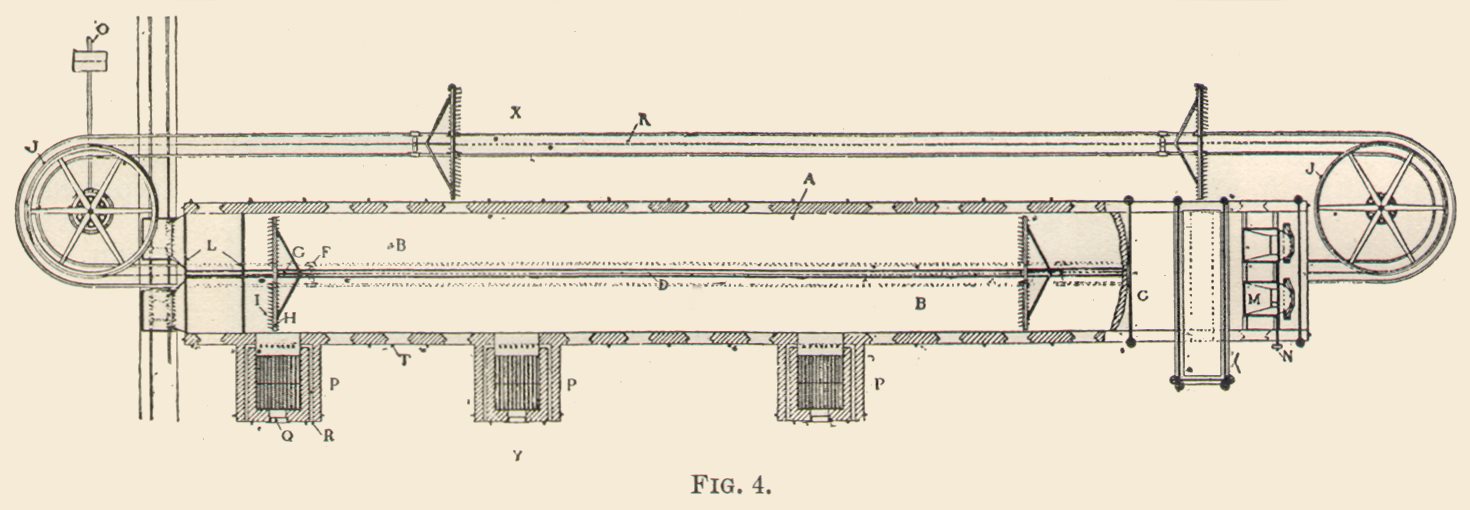
At the other end, instead of dragging in and arranging material, it skimmed off, as it were, and brought put with it, a proportion of the roasted ore, and dropped it, as it emerged, in a red-hot shower into hoppers, which led down to a small conveyor below, and that carried it to a much larger and longer water-jacketed conveyor, extending the whole length of the building. (Figs. 3 and 5.) The object of these conveyors is to cool the red-hot roasted ore on the way to the cyanide vats and hoppers over the chlorination barrels, and also to convey the material gradually to them.
The cooling conveyor, as seen in the figure, is a long, wide trough, having a water jacket underneath by which the hot ore is cooled, and down which passes a continuous series of harrow-like rabbles, armed with teeth set at different angles. These furrow and turn the hot ore, till every portion of it has been well exposed to the atmosphere and the water jacket, and cooled. At the far end of the conveyor the harrows turn over and reverse and revolve with teeth in air and pass underneath the trough; the ore falls into a chute, and is elevated to storage bins from which it is trammed to the cyanide tanks, or chlorination barrels.
When the ore is for chlorination it is discharged into the foot of an elevator by which it is carried up to the hoppers above the chlorination barrels. When the ore is for cyanide it is conveyed by the conveyor to the cyanide tanks. The Ropp furnace by the side of the conveyor is fed with oil sprayed by steam.
The cyanide vats or tanks for the most part, lie immediately below the conveyor, as shown in Figs. 3 and 5. There are eight of these cyanide vats of various sizes and capacities, the latter varying from 50 to 170 tons. One of the largest happened to be empty at the time of our visit and showed its false bottom, made of wood lattice covered with canvass and burlap. Later we saw this one in process of filling from two large pipes from the tramway overhead, which poured down a continuous shower of yellow, flour-like ore. This was leveled and distributed by a man standing in the tank. Afterwards the liquor was introduced by a pipe from below, and when it had ascended through and thoroughly pervaded the material, liquor was poured in again from above, to penetrate it by a downward percolation. The tanks are filled with liquor and material to within a few inches of the rim. These tanks or vats occupy a platform immediately below the rabble cooling conveyors. At the bottom of each tank to one side is a little gate, or aperture, through which the tailings are sluiced out with a hose, after the gold and liquor has been leached put and passed down to the room below, where are the precipitating boxes, and near them, the two storage tanks of cyanide.
The precipitating boxes stand on a bench in three sets, connected by pipes with the tanks above. The boxes slope on an inclined scale of about 10 degrees, the height of each box descending on a graduated scale to the one beneath it with a drop of an inch or so between each two boxes. Each box has a sort of lip descending nearly to the box below for the liquor to dribble over.
Each box is divided into two sections, a narrow and a wider one, by a slat which descends to within an inch of the bottom of the box allowing an aperture for the liquor to pass under and up from the narrow into the larger section, which alone contains the zinc shavings, the smaller receiving the liquor charged with gold direct from the pipe. The zinc shavings are packed in mats, one above the other, and kept down and in place on the top by a wire netting which allows also the escape of the gas generated by the chemicals.
Most of the gold is precipitated as a black slime on the shavings in the first box, the last box containing almost pure filtered liquid. From this, pipes descend through the floor to two tanks filled with nearly pure filtrated cyanide, standard 8 pounds. This liquor is pumped back to the upper tanks to be used again. What is caught finally in the riffles is the concentrates, principally pyrites. The concentrating room is the last of the series. Here are two long, shallow troughs lined with horizontal slat riffles, in two divisions, and a trough below without riffles, which leads to the final waste dump.
We may here give a brief explanation of what happens to the ore in the leaching tanks and in the filtering presses. Cyanide solutions from the leaching tanks contain free cyanide, caustic potash, gold potassium cyanide, and silver potassium cyanide and compounds of the baser metals. Gold and silver are precipitated from the solution by means of zinc shavings as a blackish powder on the zinc. The zinc itself goes into the solution. During the process hydrogen gas is evolved, and bubbles up to the surface, the bubbles being easily ignited by a match. From five ounces to one pound of zinc are required for every ounce of gold recovered. In cleaning up the zinc box the blackened, undecomposed shavings of the first box are transferred to an iron pan, the gold slime remaining on top of the screen is stirred to make it pass through, after which the screen is removed and washed.
The cock at the bottom of the compartment is opened and the slime settled there is drawn off into a bucket, where it again settles, after which the clear cyanide is decanted and returned to the zinc box. The collected slime is put on a filter, drained of cyanide and washed with water. The slime consists of metallic gold, silver, and copper precipitated from the cyanide.
In refining the precipitates, they are placed in a cast-iron pan, subjected to a dull red heat in a muffle furnace. Clean slime appears like a fine, black mud. When the roast is complete the precipitate is treated with hydrochloric acid in pans having mechanical agitators. The residue is washed and dried in filters, fluxed, and replaced in the roasting pan for drying and afterwards smelted into a bar or brick and sent to the Denver mint.
In this article we have not alluded to the chlorination department of this mill. This department, in which are treated certain ores to which this process is better adapted than the cyanide process, adjoins the other mill and presents many points of interest, but at present, space will not allow us to describe it and the other cyanide and chlorination mills, of which there are a number in the vicinity, the different processes being used according to the varying qualities of the ores.